 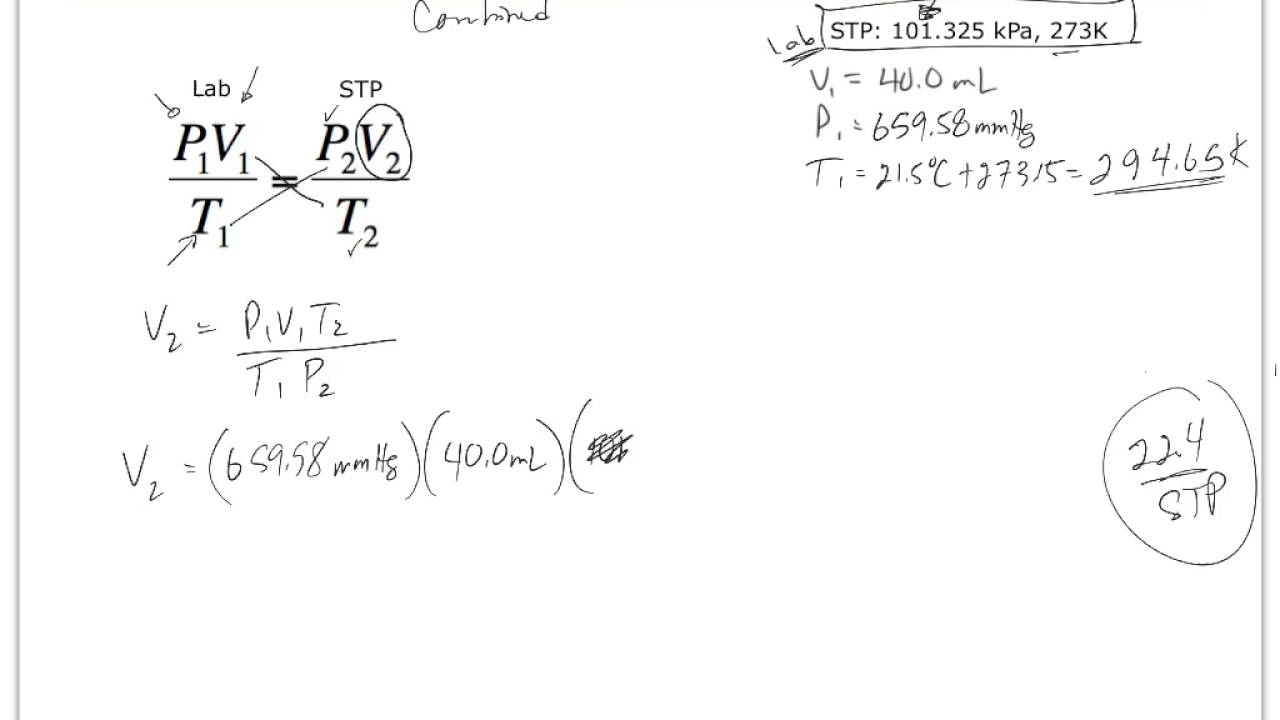
It is used in thermonuclear fusion weapons, as a tracer in isotope geochemistry, and specialized in self-powered lighting devices. Tritium has also been released during nuclear weapons tests. Trace amounts of tritium occur naturally because of the interaction of cosmic rays with atmospheric gases. It is radioactive, decaying into helium-3 through β− decay with a half-life of 12.32(2) years. (atomic mass 3.016 049 281 320(81) Da) is known as tritium and contains one proton and two neutrons in its nucleus. Main article: Tritium A tritium atom contains one proton, two neutrons, and one electron. Deuterium is also a potential fuel for commercial nuclear fusion. Heavy water is used as a neutron moderator and coolant for nuclear reactors. nuclear magnetic resonance spectroscopy. Deuterium and its compounds are used as a non-radioactive label in chemical experiments and in solvents for 1 Water enriched in molecules that include deuterium instead of protium is called heavy water. Presumably the differential concentration of deuterium in the inner solar system is due to the lower volatility of deuterium gas and compounds, enriching deuterium fractions in comets and planets exposed to significant heat from the Sun over billions of years of solar system evolution.ĭeuterium is not radioactive, and does not represent a significant toxicity hazard. Deuterium on Earth has been enriched with respect to its initial concentration in the Big Bang and the outer solar system (about 27 ppm, by atom fraction) and its concentration in older parts of the Milky Way galaxy (about 0.023%, or 23 ppm). Deuterium comprises 0.0026–0.0184% (26 ppm or 184 ppm by population, not by mass) of hydrogen samples on Earth, with the lower number tending to be found in samples of hydrogen gas and the higher enrichment (0.015% or 150 ppm) typical of ocean water. The nucleus of deuterium is called a deuteron. (atomic mass 2.014 101 777 844(15) Da), the other stable hydrogen isotope, is known as deuterium and contains one proton and one neutron in its nucleus. Main article: Deuterium A deuterium atom contains one proton, one neutron, and one electron. (see diproton for a discussion of why others do not exist) Unique among all stable isotopes, it has no neutrons. Hydrogen-1 (protium) Protium, the most common isotope of hydrogen, consists of one proton and one electron. ^ a b c Decay mode shown is energetically allowed, but has not been experimentally observed to occur in this nuclide.^ Tritium occurs naturally as a cosmogenic nuclide.^ Produced during Big Bang nucleosynthesis, but not primordial, as all such atoms have since decayed to 3.^ Produced during Big Bang nucleosynthesis.^ # – Values marked # are not purely derived from experimental data, but at least partly from trends of neighboring nuclides (TNN).Īre the only stable nuclides with more protons than neutrons.^ ( ) spin value – Indicates spin with weak assignment arguments.^ Bold symbol as daughter – Daughter product is stable.^ ( ) – Uncertainty (1 σ) is given in concise form in parentheses after the corresponding last digits.(During the early study of radioactivity, some other heavy radioactive isotopes were given names, but such names are rarely used today.) , with no neutrons, is sometimes called protium. ) instead to avoid confusion in the alphabetic sorting of chemical formulas. The IUPAC accepts the D and T symbols, but recommends using standard isotopic symbols ( 2 The symbols D and T are sometimes used for deuterium and tritium. (or hydrogen-2) isotope is deuterium and the 3 
Hydrogen is the only element whose isotopes have different names that remain in common use today: the 2 Heavier isotopes also exist, all of which are synthetic and have a half-life of less than one zeptosecond (10 −21 s). Hydrogen ( 1H) has three naturally occurring isotopes, sometimes denoted 1 The three most stable isotopes of hydrogen: protium ( A = 1), deuterium ( A = 2), and tritium ( A = 3). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
